Early trial shows promise for new prostate cancer immunotherapy
By: Dr. Avi Verma
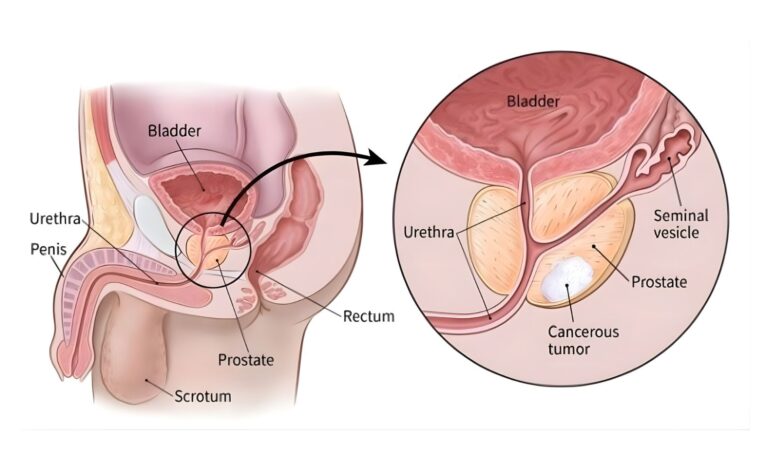
A new experimental therapy for advanced prostate cancer has generated cautious optimism among cancer researchers after early clinical trials showed encouraging results, including tumour shrinkage in some patients.
The drug, known as VIR-5500, is currently being evaluated in early-stage trials involving men whose prostate cancer continued to progress despite receiving standard treatments. Researchers say the therapy represents a new approach in the fight against one of the most common cancers affecting men worldwide.
According to global health data, around 1.4 to 1.5 million men are diagnosed with prostate cancer each year. While many cases can be successfully treated when detected early, advanced or metastatic forms of the disease remain more difficult to manage.
VIR-5500 belongs to a category of immunotherapy treatments called T-cell engagers. These therapies are designed to harness the body’s immune system by directing specialized immune cells, known as T-cells, to locate and attack cancer cells.
Scientists involved in the research explain that the drug acts as a bridge between immune cells and tumour cells. By bringing them into close contact, the immune system is able to recognize and destroy cancer cells that might otherwise evade detection.
One notable feature of the treatment is its “masked” design, which keeps the drug largely inactive in the bloodstream and activates it mainly within the tumour environment. Researchers believe this targeted mechanism may help reduce side effects while improving the drug’s ability to attack cancer cells.
Experts emphasize that the treatment remains in the early stages of development, and larger studies will be needed to confirm its effectiveness and safety before it could become widely available.
Disclaimer
This article is intended for informational and educational purposes only. The treatment mentioned is an experimental therapy currently under clinical investigation and is not yet approved for routine medical use. Patients should consult qualified healthcare professionals for medical advice, diagnosis, or treatment decisions.